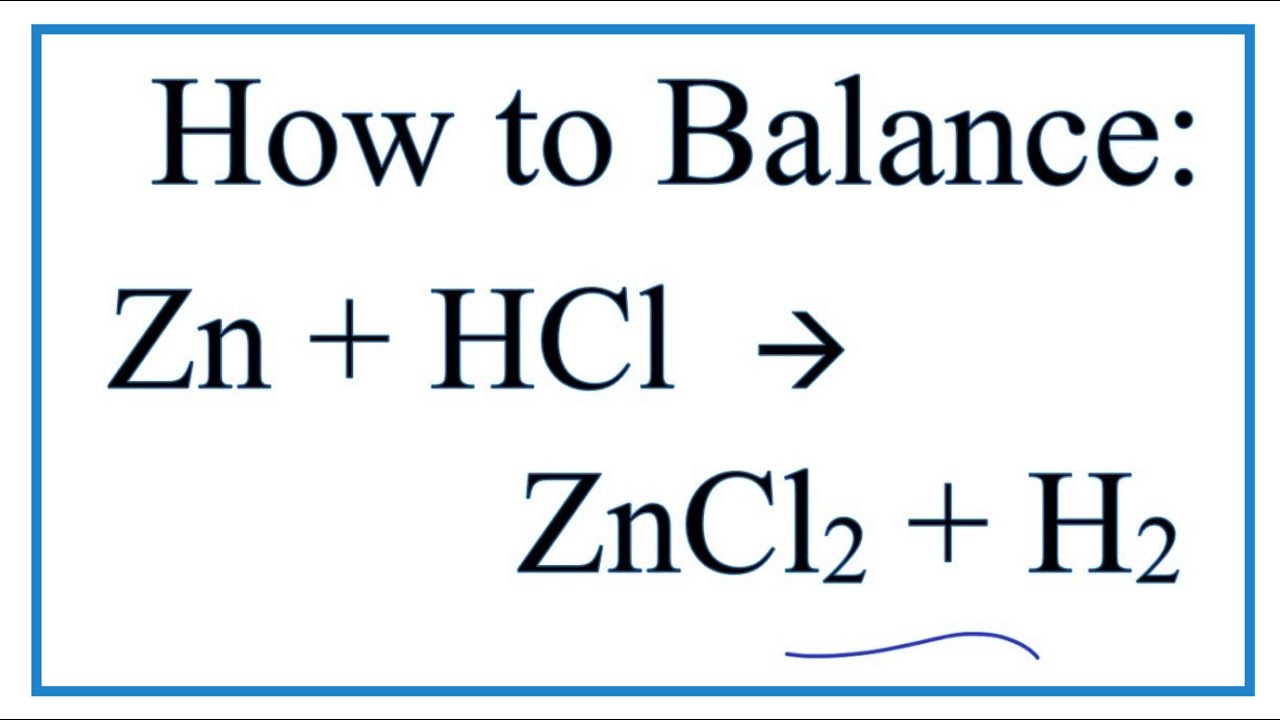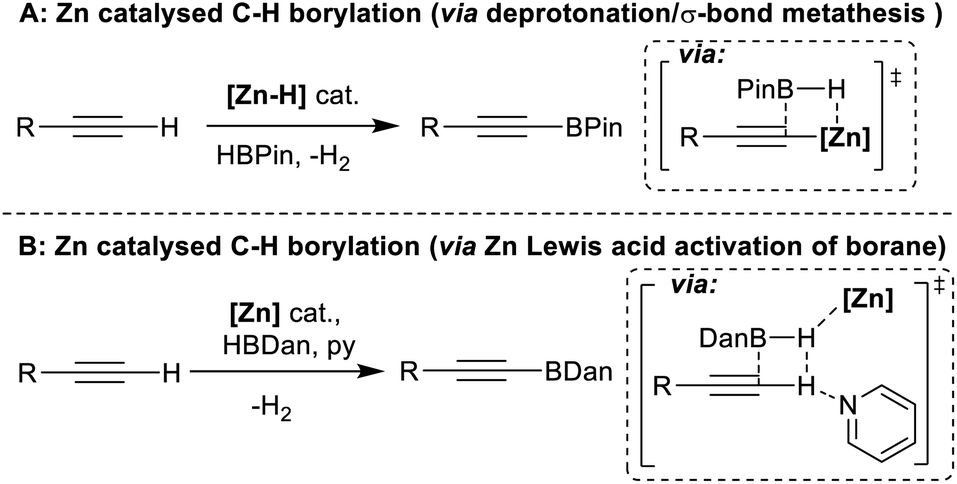
Zinc catalysed electrophilic C–H borylation of heteroarenes - Chemical Science (RSC Publishing) DOI:10.1039/D1SC01883C
![Molecular Zinc Hydride Cations [ZnH]+: Synthesis, Structure, and CO2 Hydrosilylation Catalysis - Ritter - 2020 - Angewandte Chemie International Edition - Wiley Online Library Molecular Zinc Hydride Cations [ZnH]+: Synthesis, Structure, and CO2 Hydrosilylation Catalysis - Ritter - 2020 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/8fb7eef0-0324-42ed-a2b2-135946ab80b4/anie202011480-toc-0001-m.png)
Molecular Zinc Hydride Cations [ZnH]+: Synthesis, Structure, and CO2 Hydrosilylation Catalysis - Ritter - 2020 - Angewandte Chemie International Edition - Wiley Online Library

SOLVED: In the reaction Zn + H+ —> Zn2+ + H2, Zn is oxidized and H is reduced. How many electrons would be produced and used in the balanced half-reactions? Complete the
43. Balance the following equation: Zn + (H+) —> (Zn+2) + H2 (Zinc reacts with hydrogen ion to give Zinc ion and Hydrogen gas.)

How to Balance H2SO4 + Zn = ZnSO4 + H2 (Sulfuric Acid + Zinc) | How to Balance H2SO4 + Zn = ZnSO4 + H2 (Sulfuric Acid + Zinc) Balancing equations could
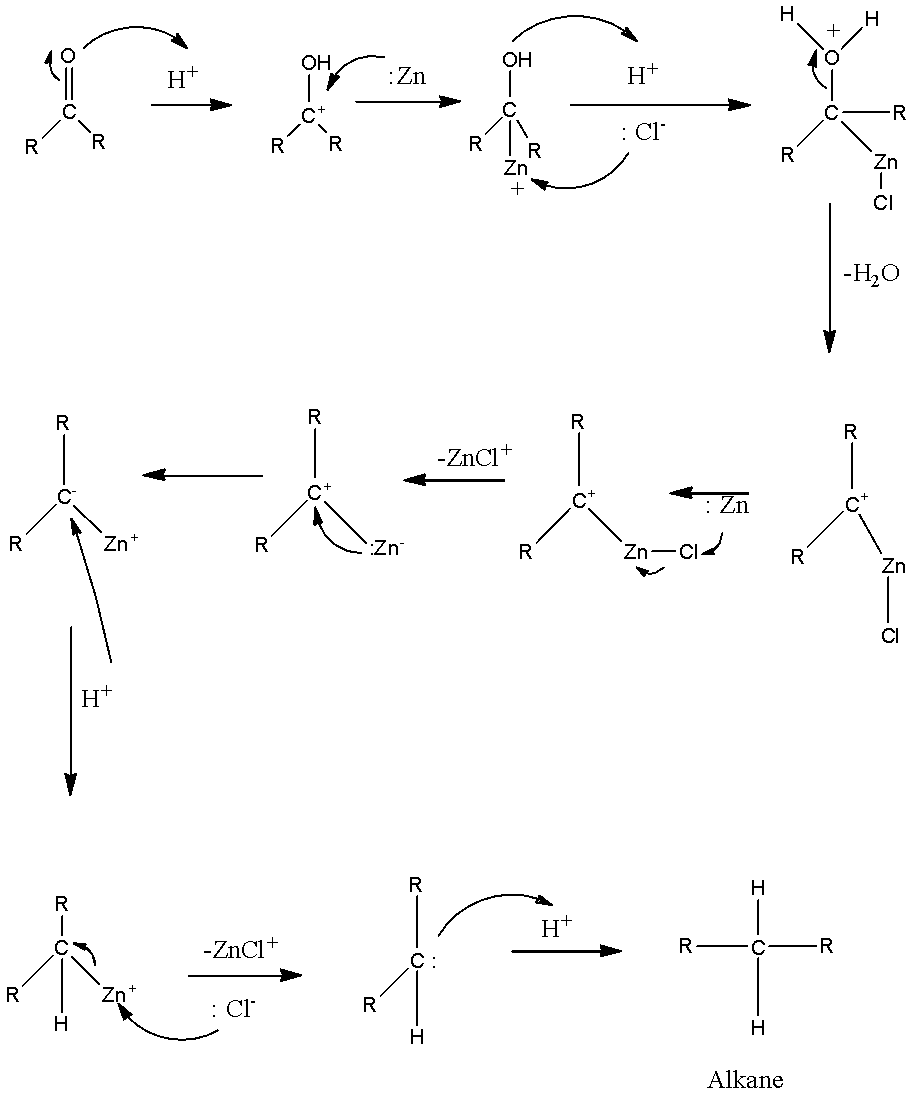
Which of the following is reduced with Zn-Hg and HCl to give alkane?(a).Ethyl acetate(b).Acetic acid(c).Acetamide(d).Butan-2-one

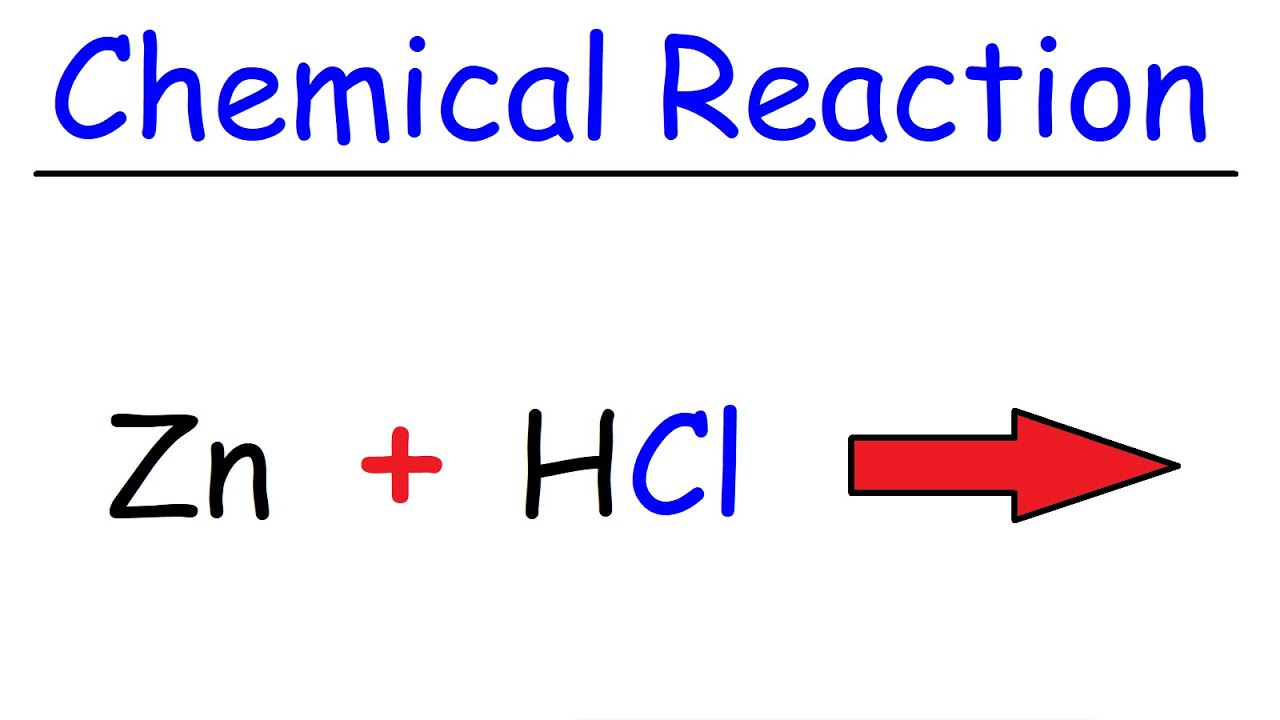
SOLVED: Consider the reaction between zinc metal and hydrochloric acid. Which of the following would increase the rate of the reaction? 3. ZnCl2 2. HCl Zn H2 using zinc powder instead of

A Comparison of Two Zinc Hydride Catalysts for Terminal Alkyne C–H Borylation/Hydroboration and the Formation of 1,1,1-Triborylalkanes by Tandem Catalysis Using Zn–H and B–H Compounds | Organometallics

Trimetallic Cu–Ni–Zn/H-ZSM-5 Catalyst for the One-Pot Conversion of Levulinic Acid to High-Yield 1,4-Pentanediol under Mild Conditions in an Aqueous Medium | ACS Catalysis
Consider the following reactions (unbalanced) Zn + hot conc. H2SO4 →G + R + X - Sarthaks eConnect | Largest Online Education Community
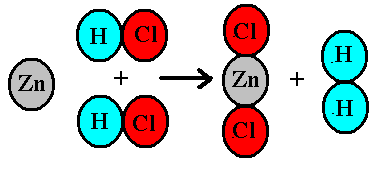

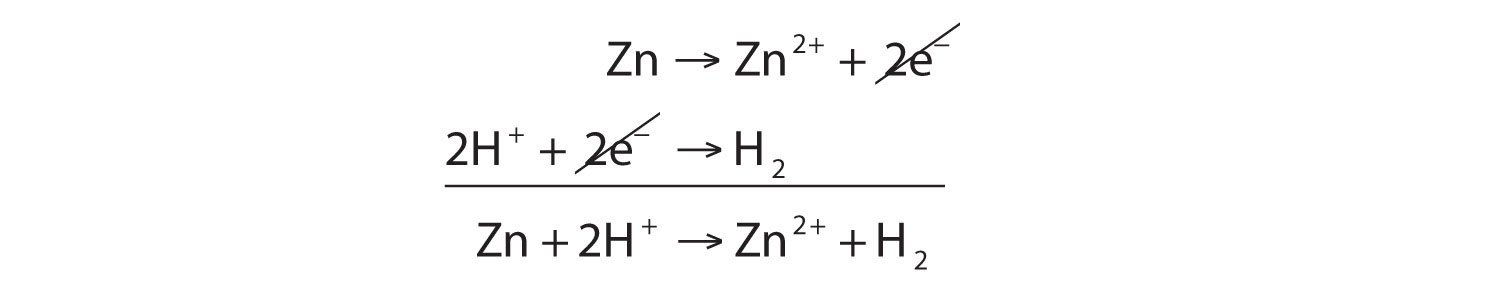

![In the reaction, C6H5COCH3 [Zn - Hg/conc. HCl][H]X . X is: In the reaction, C6H5COCH3 [Zn - Hg/conc. HCl][H]X . X is:](https://dwes9vv9u0550.cloudfront.net/images/7671796/5bdff3d8-c0c9-4fc8-89cd-f9235a0c1b30.jpg)